Model Diagram & Development
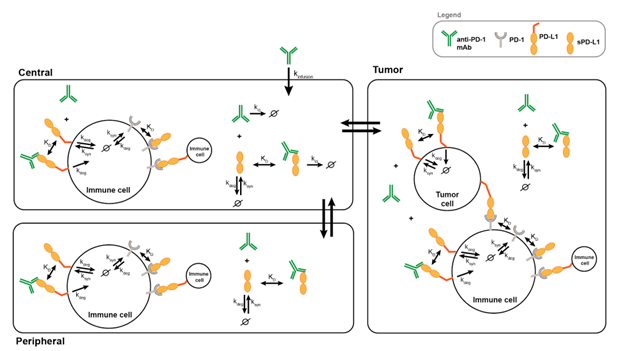
Ø indicates degradation/elimination of the indicated species in the model.
Model Description
The main reactions included in model (in all three compartments unless otherwise indicated):
- PD-L1, sPD-L1 and PD1 are constantly synthesized and degraded, and the equilibrium has been reached before dosing. • PD-1 binds to both PD-L1 and sPD-L1 reversibly.
- Drug binds to both PD-L1 and sPD-L1 reversibly.
- Drug-PD-L1 complex is degraded with the same rate constant as PD-L1.
- Drug and drug-sPD-L1 complex are eliminated in central compartment.
- Drug, sPD-L1 and drug-sPD-L1 complex transport across the compartments.